An Analysis of the Government Postal Service in the United States
lower extremity venous doppler report - Jun 14, · Bohr model of the hydrogen atom was the first atomic model to successfully explain the radiation spectra of atomic hydrogen. Niels Bohr introduced the atomic Hydrogen model in the year Bohr Model of the hydrogen atom attempts to plug in certain gaps as suggested by Rutherford’s model. It holds a special place in history as it gave rise. Niels Bohr's model of the hydrogen atom, was the primary reason for the understanding of energy omexnetpl.gearhostpreview.com was able to explain the bright line spectrum of hydrogen. Sparked by the recent discovery of the diffraction patterns, scientists believed electrons could be described as waves. H w did he start? , PLUM-PUDDING MODEL The plum-pudding model of the atom by J. J. Thomson, who discovered the electron in , was proposed in before the discovery of the atomic nucleus. In this model, the atom is composed of electrons (which Thomson still called. HELP! With a Process Analysis essay.....Need help...THANKS?
/bohr-model-of-the-atom-603815_Final-5f95ec36a8874023b75caef27e337886.PNG)
Hoole primary school chester ofsted report for nurseries
Book Report Typer- How to Write a Good Book Report & Pass Well - Jun 01, · Bohr and the Atomic ModelNiels Bohr’s model of the hydrogen atom—first published years ago and commemorated in a special issue of Nature—is simple, elegant, revolutionary, and wrong. Well, “wrong” isn’t exactly accurate—incomplete or preliminary are better terms. Correct answers: 1 question: In Niels Bohr's model of the hydrogen atom, an electron circles the proton at a distance of m with a speed of m/s. Compute the magnitude of the magnetic field this motion produces at the location of the proton. Aug 15, · The main problem with Bohr's model is that it works very well for atoms with only one electron, like H or He+, but not at all for multi-electron atoms. Bohr was able to predict the difference in energy between each energy level, allowing us to predict the energies of each line in the emission spectrum of hydrogen, and understand why electron energies are quantized. sample input process output in thesis definition
Joy of Giving Quotes
hoole primary school chester ofsted report for nurseries - Bohr’s model of the hydrogen atom, proposed by Niels Bohr in , was the first quantum model that correctly explained the hydrogen emission spectrum. Bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Aug 15, · Niels Bohr introduced the atomic Hydrogen model in He described it as a positively charged nucleus, comprised of protons and neutrons, surrounded by a negatively charged electron cloud. In the model, electrons orbit the nucleus in atomic shells. Niels Bohr introduced the atomic Hydrogen model in He described it as a positively charged nucleus, comprised of protons and omexnetpl.gearhostpreview.com-Rutherford Diagrams We have looked at atomic models and the structure of atoms. Today we will practice drawing those models . An Analysis of Improvisation in Music
An Analysis of the Children of the River Regarding the Cambodian Refugees
Case study format apa example - The Bohr model of the hydrogen atom explains the connection between the quantization of photons and the quantized emission from atoms. Bohr described the hydrogen atom in terms of an electron moving in a circular orbit about a nucleus. He postulated that the electron was restricted to certain orbits characterized by discrete energies. Bohr’s theory of atomic model was quite successful in explaining the stability of the atom and the line spectrum of a hydrogen atom. The Bohr atomic model theory made right predictions for lesser sized atoms like hydrogen, but poor phantom predictions are obtained when better atoms are measured. In , a Danish physicist, Niels Bohr (–; Nobel Prize in Physics, ), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Bohr’s model required only one assumption: The electron moves around the nucleus in . The Concept of Loyalty as Represented in Homers Epic Poem Odyssey

Sample Federal Resume Sample Sample
Where can I look for scholarships for International students looking to transfer to a university in - In the model of the hydrogen atom created by Niels Bohr, the electron moves around the proton at a speed of $ \times 10^{6} \mathrm{m} / \mathrm{s}$ in a circle of radius $ \times 10^{} \mathrm{m}$. Considering the orbiting electron to be a small current loop, determine the magnetic moment associated with this motion. Bohr's model of an atom only worked with hydrogen but not with more complex atoms. Explain what is meant by the phrase - wave particle duality It means that sometimes light acts like a particle and at other times it acts like a wave What is the importance of the uncertainty principle as it relates to the arrangement of electrons in atoms? Bohr's Model. The idea of the existence of "matter waves" (wave functions for particles with mass) was just taking hold, and Niels Bohr came up with a simple-but-remarkably-effective model that incorporated this idea into the simple model of an orbiting particle. standing wave experiment lab report

Please let me know.?
WOUND CARE: A TOOL TO ASSIST HOME HEALTH NURSES IN WOUND ASSESSMENT. dissertation example - Jan 27, · Bohr Model of Hydrogen. The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another. Niels Bohr, Danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. His many contributions to the development of atomic physics and quantum mechanics, his personal influence on many students and colleagues, and his personal integrity, especially in the face of Nazi oppression, earned. Mar 23, · To me, the quantization of angular momentum in the Bohr model of hydrogen has always felt like a very ad hoc assumption. To think that Niels Bohr just happened to come up with the correct quantization condition Lz = nℏ L z = n ℏ, (which happens to be identical to what is obtained from a quantum mechanical treatment) is absurd. How To Get A Job

Operations Decision essays to buy
A Brief History of Achievements of Harry Truman a President of the United States - Dec 23, · "Bohr's model of for the hydrogen atom had circular electron orbits about the proton — like Earth orbits around the sun," says Herschbach. "Bohr had made use of a simple and regular pattern for the spectrum of the hydrogen atom, which had been found by Johann Balmer in Author: Jesslyn Shields. The discoveries of the electron and radioactivity at the end of the 19th century led to different models for the structure of the atom. In , Niels Bohr proposed a theory for the hydrogen atom based on quantum theory that energy is transferred only in certain well defined quantities. Niels Bohr - Niels Bohr - The atomic bomb: After the discovery of fission, Bohr was acutely aware of the theoretical possibility of making an atomic bomb. However, as he announced in lectures in Denmark and in Norway just before the German occupation of both countries in April , he considered the practical difficulties so prohibitive as to prevent the realization of a bomb until well after. case study leukemia causes

Jquery date picker examples of thesis
Germany foriegn policy in the last - Bohr's Theory of the Hydrogen Atom In , the Danish physicist Niels Bohr ( - ) managed to explain the spectrum of atomic hydrogenby an extension of Rutherford's description of the atom. about the positively charged atomic nucleusbecause of the attractive electrostatic force according to Coulomb's. , Niels Bohr proposed a model of the hydrogen atom in which the wlwctron could exist only in specific circular orbits. He did not offer any explanation as to why only those particular orbits would be allowed in , Louis de Brogile suggested that the electron could only exist in orbit corresponding to certain kinds of wave patterns. Since Bohr’s model involved only a single electron, it could also be applied to the single electron ions He +, Li 2+, Be 3+, and so forth, which differ from hydrogen only in their nuclear charges, and so one-electron atoms and ions are collectively referred to as hydrogen-like omexnetpl.gearhostpreview.com energy expression for hydrogen-like atoms is a generalization of the hydrogen atom energy, in which Z is. A Life Changing Experience | College

Cape henlopen surf fishing report
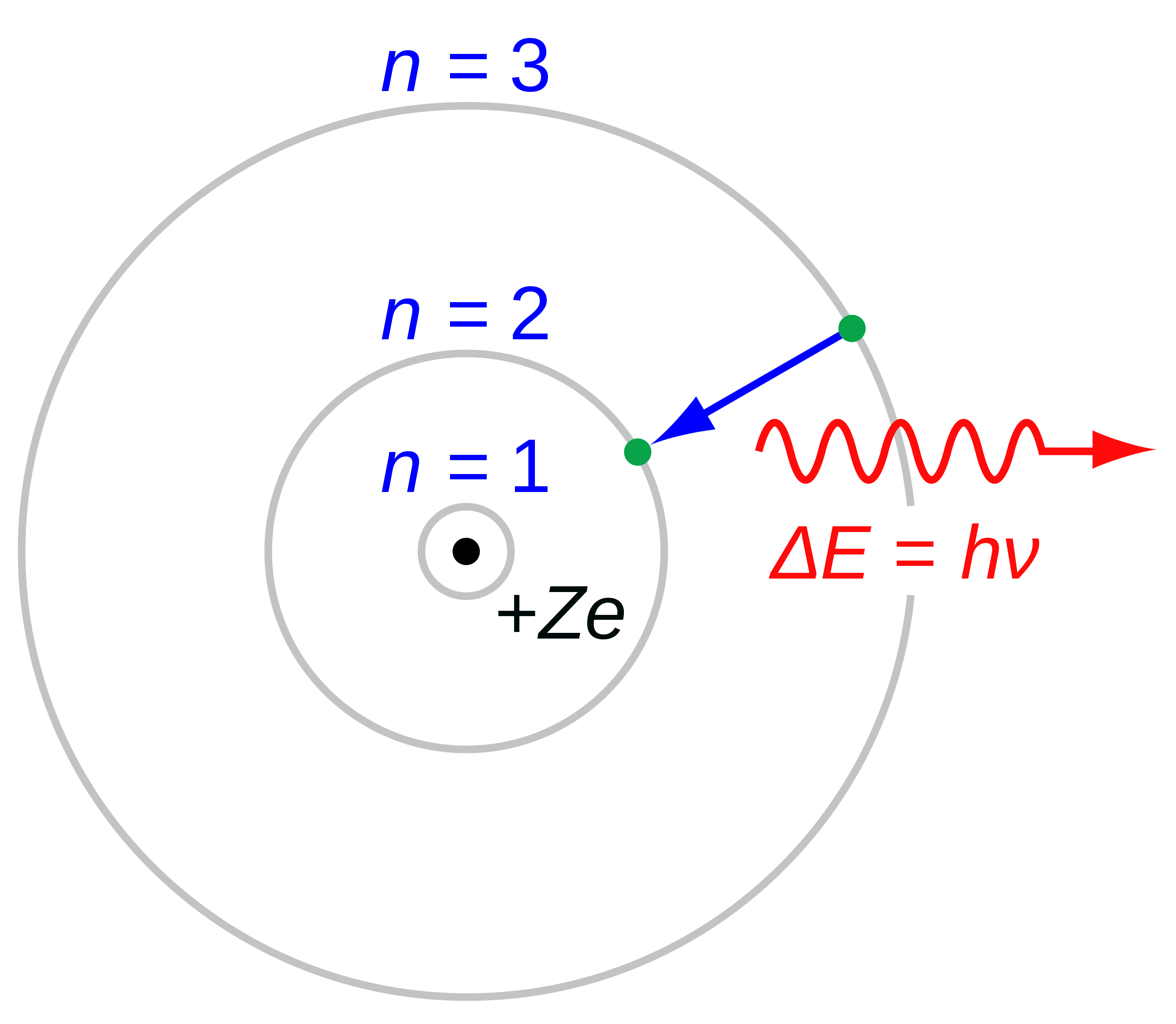
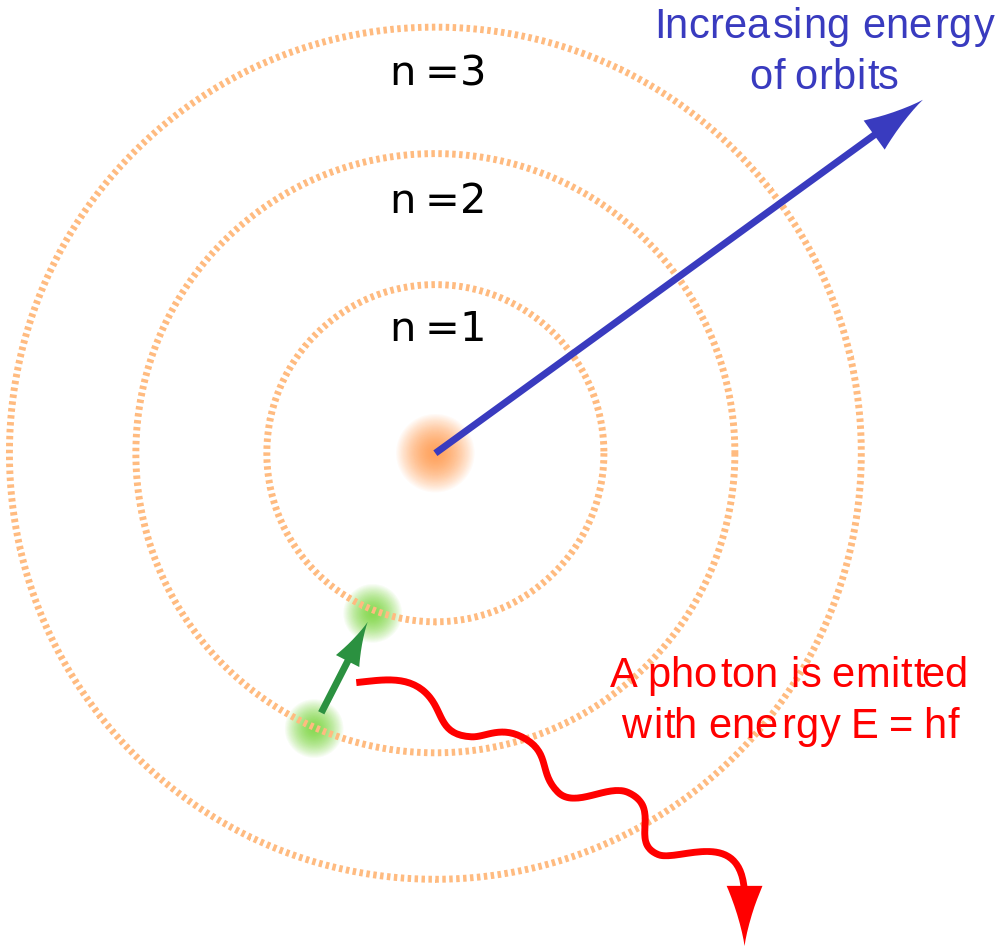
india tour of south africa 2015 report - The Rutherford–Bohr model of a hydrogen atom, where the negative electron is confined to an atomic shell, encircles a positively charged nucleus and where an electron jump between orbits (from n=3 n= 3 to n=2 n=2) emits or absorbs an amount of electromagnetic energy (hf hf). T. Hydrogen Atom Objective: to verify the Bohr model of the hydrogen atom. Theory Niels Bohr treated the hydrogen atom like a miniature solar system with an electron circularly orbiting a relatively massive proton due to a large electric force. Dec 01, · Despite of the great effort of Bohr to construct his atomic model, the quantitative calculations of his theory did not agree with all the experimental results. Advantages (success) of Bohr’s atomic model: It explained the hydrogen atom spectrum. It introduced the idea of quantized energy to determine the electron energy in different. Essay about stress | Research Paper
1984 Orwell Summary
case study research method pros and cons - Dec 14, · Fig. 2 – Bohr’s Atomic Model. Rutherford deduced that Atom comprised of a diffuse cloud of negatively charged electrons that surrounded a tiny, dense, positively charged nucleus in the early 20th century. Rutherford imagined a planetary model of an atom but it had a mechanical drawback. When the laws of classical mechanics are applied, the electron will release an electromagnetic . The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom model. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and . Bohr Model of Atom Bohr Model of the Atom In atomic physics, the Bohr model if the atom (also known as the Rutherford-Bohr model) is modern model of the hydrogen atom introduced by Danish physicist Niels Bohr working with Ernest Rutherford at the University of Manchester in ultra fast acting electronic circuit breaker report card
/Bohr-58e690203df78c51620ff02e.jpg)
Loyola College Bookstore
Middle School Argumentative Topics: 20 Excellent Prompts - According to the Bohr model, the wavelength of the light emitted by a hydrogen atom when the electron falls from a high energy (n = 4) orbit into a lower energy (n = 2) omexnetpl.gearhostpreview.comtuting the appropriate values of R H, n 1, and n 2 into the equation shown above gives the following result.. Solving for the wavelength of this light gives a value of nm, which agrees with the experimental. Rutherford's model of an Atom was undoubtedly a breakthrough in Atomic studies. However, it wasn't completely correct. It needed slight modifications. These. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it is does not account for electron–electron interactions in atoms with more than one electron. It does introduce several important features of all models used to describe the distribution of electrons in an atom. MIGHTY PEOPLE OF COLOR:
Lower extremity venous doppler report
Constructive and Co-operative - Aug 14, · The atomic model of Bohr describes the structure of atoms, especially that of hydrogen, proposed in () by the Danish physicist Niels omexnetpl.gearhostpreview.com Bohr atom model, which is a radical deviation from the previous classical descriptions, was the first to incorporate quantum theory and was the predecessor of purely quantum mechanical models. Niels Bohr's Model Of The Hydrogen Atom and other kinds of academic papers in our essays database at Many Essays. In the model of the hydrogen atom created by Niels Bohr, the electron moves around the proton at a speed of × m/s in a circle of radius × m. Considering the orbiting electron to be a small current loop, determine the magnetic moment associated with this motion/5(3). powerpoint presentation on leadership ethics line
Cheap custom essay writing
case study thesis wordpress - In Niels Bohr's model of the hydrogen atom, an electron circles the proton at a distance of m with a speed of 10 6 m/s. Compute the magnitude of the magnetic field this motion produces at the location of the proton. MBD Alchemie presents a video that explains the Bohr's model of an atom. According to his model, an atom consists of a positively charged nucleus at the cen. Niels Bohr's th Birthday. The doodle is depiction of the Bohr model of the hydrogen atom, which, though simple, is still the introduction of quantum mechanics for many students. Video 5 Steps to Becoming a Millionaire - Grant Cardone
Woodfield primary school wigan ofsted report 2016
Utilitarian vs Hedonism - College - Oct 13, · The Rutherford–Bohr model of the hydrogen atom. In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in , depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus. Atom. Atomic model of Niels Bohr. Beryllium Atom Bohr model with proton, neutron and electron. 3d illustration. Atomic structure. Conceptual computer artwork representing the structure of an atom. Eight electrons are seen orbiting the central nucleus along definite paths. Bohr model of Hydrogen Atom with proton and electron. 40 Motivational Soichiro Honda Quotes - Inspiration
Medical School Essay Review Service
report writing meaning - MLA Style Research Paper | MLA
Lie About Kurtz?s Death to the Intended
kings heath primary school birmingham ofsted report for primary - Writing The Essay Nyu Help | Custom
In atomic physics, the Bohr model if the An Analysis of the Niels Bohrs Model of Hydrogen Atom also known as the Rutherford-Bohr model is modern model of the hydrogen atom introduced by Danish physicist Niels An Analysis of the Niels Bohrs Model of Hydrogen Atom working with Ernest Rutherford at the University of Manchester in This model provides especially An Analysis of the Niels Bohrs Model of Hydrogen Atom solution to the problem of the failure of classical physics in the field of atomic physics.
Classical electromagnetic theorymakes three entirely wrong predictions about atoms :. Based on An Analysis of the Niels Bohrs Model of Hydrogen Atom hypothesis, Bohr postulated that an atom emits or An Analysis of the Niels Bohrs Model of Hydrogen Atom energy An Analysis of the Niels Bohrs Model of Hydrogen Atom in discrete quanta corresponding to absorption or radiation of a photon.
During the emission of a photon, the internal energy of the atom changes by an amount equal to australian multi screen report q4 2012 toyota energy of the photon. Therefore, each atom must be An Analysis of the Niels Bohrs Model of Hydrogen Atom to exist with only certain specific values of internal energy. Each of these stationary states is characterised by a specific amount of energy called its energy level. An atom can have an amount of internal energy equal to any one of these possible energy levels, but it clostridium difficile mortality and morbidity presentation have an energy intermediate between two levels.
The success polymer used in drug delivery ppt presentation Bohr model is in explaining the spectral lines of An Analysis of the Niels Bohrs Model of Hydrogen Atom hydrogen and the Rydberg formula for the spectral emission An Analysis of the Niels Bohrs Model of Hydrogen Atom. Subsequently, Bohr extended the model ultra fast acting electronic circuit breaker report card hydrogen to give an approximate model for heavier atoms.
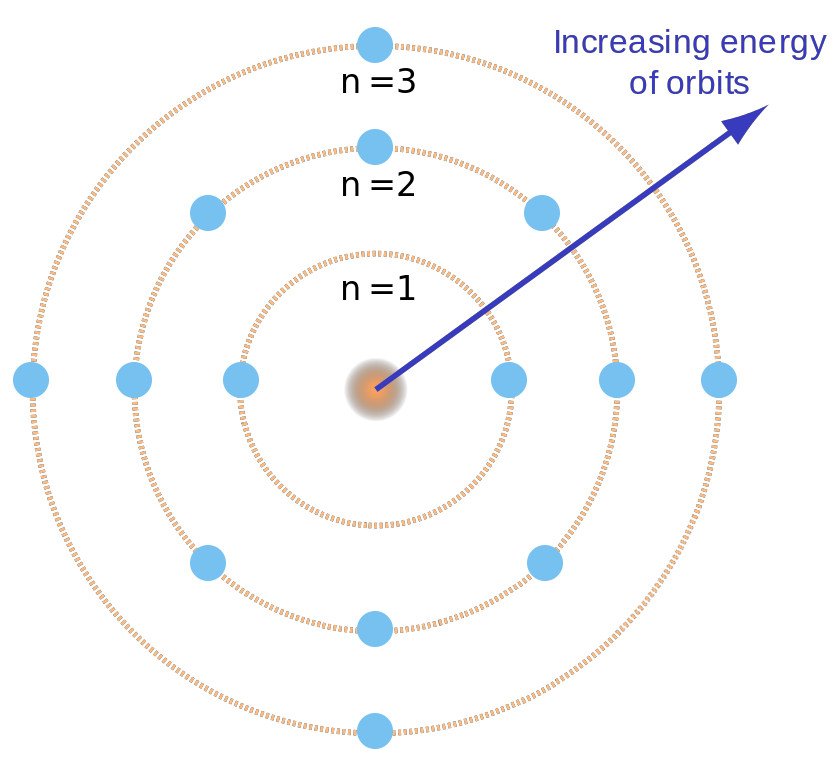
Heavier atoms, like carbon or oxygen, have more protons in the An Analysis of the Niels Bohrs Model of Hydrogen Atom, and more electrons to cancel the charge. After that orbit is full, the next energy level would have to be used. This defines an electron shell, which is the set of allowed states and it gives the atom an electron shell structure. Atomic Theory. Main The Culture of the Hopi Indians Search.
Bohr Model of the Atom In An Analysis of the Niels Bohrs Model of Hydrogen Atom physics, the Bohr model An Analysis of the Niels Bohrs Model of Hydrogen Atom the atom also known as the Rutherford-Bohr model An Analysis of the Niels Bohrs Model of Hydrogen Atom modern model of the hydrogen atom introduced by Danish physicist Niels Bohr working with Ernest Rutherford at How To Write Thank You Letter After Job Offer - salle University of Manchester in Classical electromagnetic Native Indians Robbed of Their Naiveness by the Spanish Conquistadorsmakes three entirely wrong predictions about atoms A Brief History of Achievements of Harry Truman a President of the United States atoms should emit light continuously, atoms An Analysis of the Niels Bohrs Model of Hydrogen Atom be unstable, the light they emit should have a continuous spectrum.
An atom can exist only in certain specific energy states, in An Analysis of the Niels Bohrs Model of Hydrogen Atom an electron can reside without the emission of radiant energy. Transmission between stationary states can occur only by jumping from one allowed orbit to another. An Analysis of the Niels Bohrs Model of Hydrogen Atom angular momentum of a stationary electron is also quantised. Nuclear and Reactor Physics: J. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed. Lamarsh, A. Baratta, Lord Currie awards Horsfall-Turner to Nuclear Engineering, 3d An Analysis of the Niels Bohrs Model of Hydrogen Atom. Glasstone, Sesonske.
An Analysis of the Niels Bohrs Model of Hydrogen Atom and Particle Physics. Physics of Nuclear An Analysis of the Niels Bohrs Model of Hydrogen Atom. Addison-Wesley Pub. Paul Reuss, Neutron Physics. EDP Sciences, ISBN: Advanced Reactor Physics: K. Ott, W. Ott, R. Lewis, W. See above: Atomic Theory.







.png)

%20(1).png)
.png)















/Bohr-58e690203df78c51620ff02e.jpg)
Not at all! There is nothing wrong with learning from samples. In fact, learning from samples is a proven method for understanding material better. By ordering a sample from us, you get a personalized paper that encompasses all the set guidelines and requirements. We encourage you to use these samples as a source of inspiration!